FTC Announces Annual Update to HSR and Section 8 Thresholds
Client Alert | 1 min read | 01.20.26
The Federal Trade Commission (FTC) has announced its annual updates to the thresholds and filing fees related to the Hart-Scott-Rodino Antitrust Improvements Act of 1976 (the HSR Act). These dollar thresholds are indexed annually based on changes in the U.S. gross national product and the Consumer Price Index.
The HSR Act requires that certain transactions be notified prior to their consummation. This year, the “size-of-transaction” threshold for reporting mergers and acquisitions under the HSR Act will increase from $126.4 million to $133.9 million. In addition, the “size-of-person” threshold, the filing fee thresholds, and the fee schedule will all also increase. The new filing fee thresholds and fee schedule are as follows:
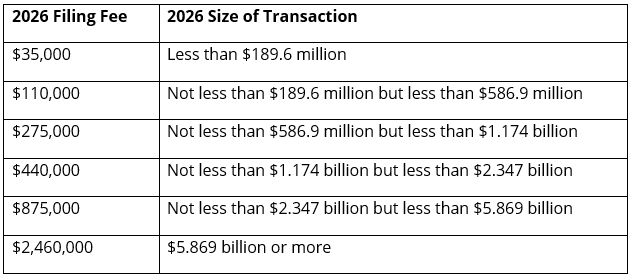
The FTC also issued revised thresholds relating to the prohibition of certain interlocking directorates under Section 8 of the Clayton Act.
The revised thresholds will become effective 30 days after their publication in the Federal Register, which is expected in the next few days. Click here to read a full copy of the FTC's announcement, including a complete listing of the revised thresholds. Click here for a copy of the FTC’s announcement and information regarding the Clayton Act, Section 8 thresholds.
We would like to thank Associate Emil Zhang for their contribution to this alert.
Contacts
Insights
Client Alert | 5 min read | 06.01.26
California Court Upholds Insurer’s Duty to Defend After Covered Claim Is Dismissed
On April 30, 2026, the U.S. District Court for the Northern District of California issued a significant ruling in an insurance coverage dispute between a commercial general liability insurer and its policyholder. The decision addresses several critical issues in insurance law, including the scope and continuity of the duty to defend and the standard for insurer reimbursement of defense costs in mixed-claim actions. The court ruled largely in favor of the insured, SVO Building One, LLC ("SVO"), and the matter now heads toward settlement or trial on SVO's remaining counterclaims.
Client Alert | 2 min read | 05.29.26
California Assembly Passes AB 1776, Sending Major Antitrust Bill to the Senate
Client Alert | 5 min read | 05.29.26
Clover Insurance v. HHS: S.D. of Georgia Holds 20 Star Ratings Measures Unlawful
Client Alert | 3 min read | 05.29.26






